Language Learning in Children: Vocabulary Acquisition in a Meaningful Context
Do you have a 5 to 10 year old child with Fragile X syndrome? Participate in this remote research study and help researchers understand how children learn words. Researchers at the Neurodevelopmental Language and [...]
Lunch & Learn Series: Single-Dose Medication Study Results in Fragile X Syndrome
Dr. Craig Erickson joined us for a 45-minute webinar where he presented the results of a single-dose medication study in FXS and then answered questions during a moderated Q&A.
Fragile X Program at Thompson Autism and Neurodevelopmental Center, Children’s Hospital of Orange County
Learn more about the CHOC Fragile X Program, what to expect when you visit, and meet Drs. Sailaja Golla and J. Thomas Megerian.
Study: Recording the Experiences of Black and Hispanic Mothers who have Children with Fragile X Syndrome
Are you a Black or Hispanic woman raising a child with Fragile X syndrome? Researchers want to hear about your experiences. The South Carolina Family Experiences Lab is conducting 30-minute interviews to learn about [...]
The National Fragile X Foundation — The Early Years: 1984–1990s
The focus in the early years of the NFXF was to “get the word out!” During that time period, the inherited nature of Fragile X was not fully understood. However, early pioneers in the Fragile X world, such as Dr. Stephanie Sherman and Dr. Ted Brown, were steadily making progress in sorting out the genetics.
2024 NIH Fragile X Centers of Excellence — Webinar
Hear updates about the NIH-funded Fragile X Centers of Excellence, which support research to improve the diagnosis and treatment of Fragile X syndrome and its related conditions.
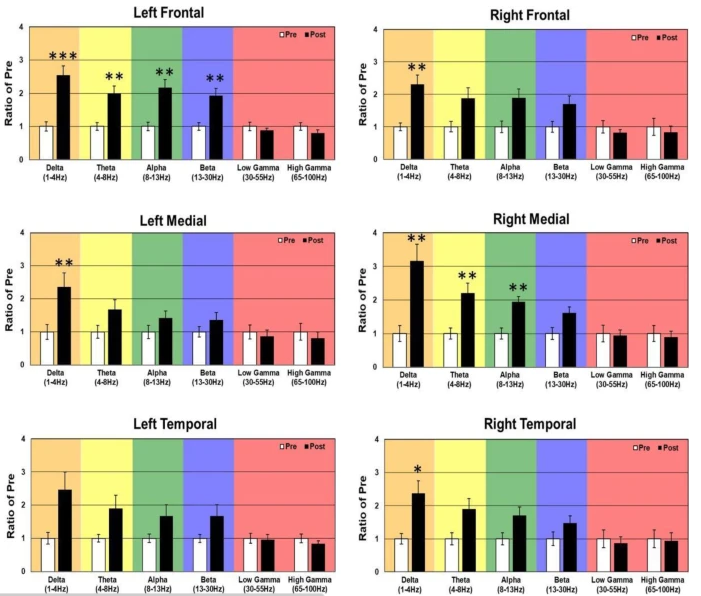
Baclofen-associated neurophysiologic target engagement across species in Fragile X syndrome
Authors: Carrie R. Jonak , Ernest V. Pedapati, Lauren M. Schmitt, Samantha A. Assad , Manbir S. Sandhu, Lisa DeStefano, Lauren Ethridge, Khaleel A. Razak, John A. Sweeney , Devin K. Binder and Craig [...]
Introductory Discussion on Estate Planning — Webinar
We hosted an introductory discussion on bequests, wills, trusts, life insurance, and IRA transfers. The panel provided insight on navigating the complexities of wealth transfer, ensuring your legacy is preserved and your loved ones are well cared for.
The National Fragile X Foundation — 1984: The Beginning
At the beginning of the 1980s, a young developmental pediatrician, Dr. Randi Hagerman, was building a career at Children’s Hospital Colorado. Her curiosity led her to some of the early papers on X-linked intellectual disabilities (in particular, those of Dr. Gillian Turner from Australia) and descriptions of what was still often referred to as Martin-Bell Syndrome.
Making Waves for Fragile X: The 7th Annual Fishing for a Cure Fundraiser
The annual Fishing for a Cure fundraiser, hosted by Joey Christoff in honor of his son Mitchell, was another great success in 2023! Check out the big fish and the big smiles.
Healthcare Experiences of African American Women with a Fragile X Premutation
Authors: Andy King, Nadia Ali, Cecelia Bellcross, Fabienne Ehivet, Heather Hipp, Jessica Vaughn, Emily G. Allen An estimated 1 in 291 women carry a Fragile X premutation (PM) and there is little evidence that [...]
Emotion Dysregulation in Fragile X Syndrome
By Mya Jones Authors: Rebecca C Shaffer, Debra L Reisinger, Lauren M Schmitt, Martine Lamy, Kelli C Dominick, Elizabeth G Smith, Marika C Coffman, Anna J Esbensen Summary: A large portion of individuals with Fragile [...]