Authors: Carrie R. Jonak, Ernest V. Pedapati, Lauren M. Schmitt, Samantha A. Assad, Manbir S. Sandhu, Lisa DeStefano, Lauren Ethridge, Khaleel A. Razak, John A. Sweeney, Devin K. Binder & Craig A. Erickson[1]
Read the full article from the Journal of Neurodevelopmental Disorders
Summary
Fragile X syndrome (FXS) is the most common inherited form of neurodevelopmental disability and the most common single gene cause of autism spectrum disorder (ASD). FXS is often characterized by intellectual disability, anxiety, repetitive behavior, social communication deficits, delayed language development. Using electroencephalogram (EEG) technology these researchers and others have identified abnormal sensory processing in FXS including hypersensitivity and reduced habituation (adaptation) to repeated stimuli. An EEG is an instrument used to measure electrical activity in the brain using small discs (called electrodes) that attach to a cap worn on the scalp.
In this study, researchers identified EEG biomarkers that are conserved between the mouse model of FXS (referred to as “FMR1 KO mice”) and humans with FXS. Biomarkers are measures that show what’s going on in the body at a given moment; EEG biomarkers are measures that capture what’s happening in certain areas of the brain at specific points in time. In sum, researchers identified brain activity in FMR1 KO mice that they also observed in humans with FXS before and after a single-dose of drug therapy.
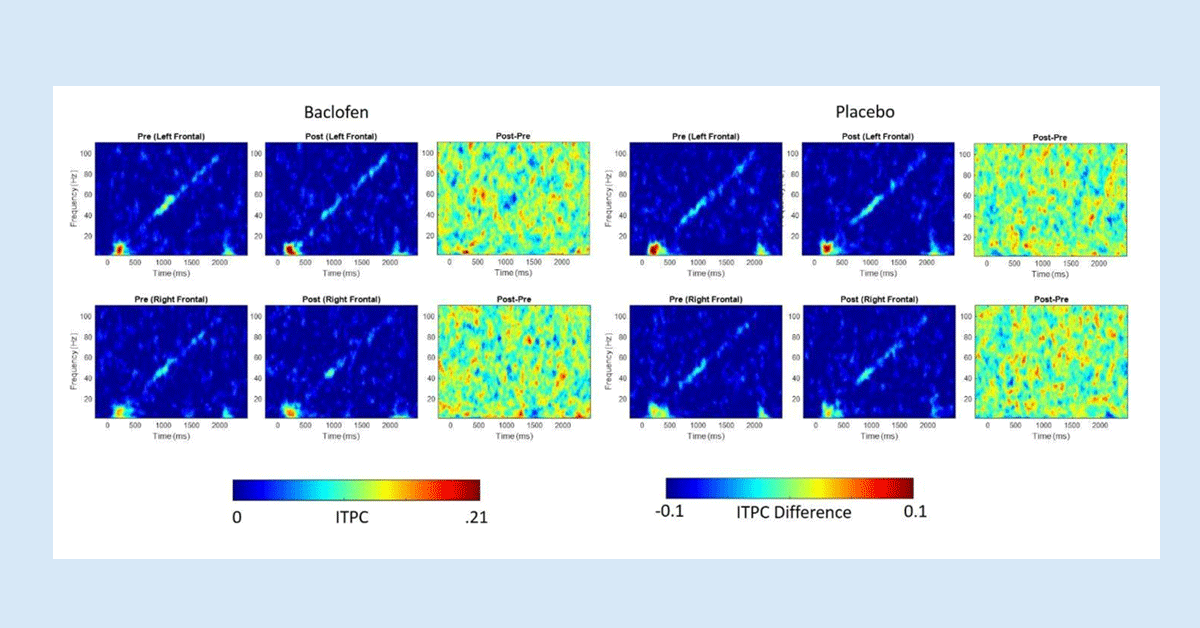
The drug therapy that researchers used in this study was a drug called racemic baclofen (RBAC). RBAC works on the brain’s GABA receptors. GABA receptors play an important role in things like regulating emotion and anxiety, amongst other things. The researchers administered one single dose of RBAC to FMR1 KO mice and to humans with FXS. Researchers then analyzed the brain activity and the effects of RBAC on the mice and the human participants by conducting EEG tests at specific timepoints both pre- and post-RBAC administration. In FMR1 KO mice and in humans with FXS, RBAC use was associated with suppression (lowering) of elevated gamma power and increase in low-frequency power at rest. Overall, researchers noted corresponding RBAC engagement on the brain’s physiology at rest across species in FXS. This means that researchers saw agreeing results from the drug in both mice and humans!
Why This Matters
Improving the translation and synchronization of outcome measures (endpoints researchers pick to assess the effect of an intervention or treatment) across animals and human beings is an essential element to the success of drug development in the FXS field as well as other neurodevelopmental disorder fields. Research studies and clinical trials must follow strict guidelines, starting with laboratory science, then moving into animal models, and eventually reaching their targeted human population. Yet, many new therapeutics that show promise in animal models, like FMR1 KO mice, have failed in clinical trials in humans with FXS due to the major differences in our two species.
The results found from the EEGs in both the FMR1 KO mice and humans with FXS are extremely important for the future of FXS research because they show a measurable commonality between the two very different species. The findings of this study hold promise for future translational medicine approaches to drug development for FXS. We can continue to further validate these results and later adjust future research in this field to incorporate these EEG biomarkers.
Next Steps
The identification of comparable biomarkers in humans and validated animal models is a critical step in facilitating pre-clinical to clinical therapeutic pipelines. This step will help to advance treatment development for neurodevelopmental disorders. More studies are needed to validate these results. More work is required, specifically to clarify dose-response patterns in beneficial and potentially harmful effects, and to identify potential baseline EEG characteristics, molecular aspects of FXS, sex, age, and other features that predict RBAC response, as such findings would help individualize patient care and potentially stratify patients in clinical trials.
Synchronizing pre-clinical (non-human) and human study will create a model that can lessen risks of large-scale clinical trials and drug development programs in this field, which will, in turn, help to guide more optimal and informed decisions during treatment development efforts.
Funding
This work was supported by National Institutes of Health grants U54 HD104461 NICHD, NINDS and U54 HD082008 NICHD, NIMH.