NFXF Data Repository
Qualitative Survey Data Now Available!
The NFXF Data Repository was created to help advance Fragile X research by making previously inaccessible, collected data available to researchers and investigators. By centralizing data from caregiver surveys, research studies, and other data collection sources, the NFXF Data Repository streamlines data access for the purposes of accelerating effective treatments and improving the quality of life for individuals living with Fragile X.
Learn more about the NFXF Data Repository.
1. Available Datasets
There are currently three available datasets, and you can learn more about each below.
- Anxiety Survey — Observable Symptoms of Anxiety in Individuals with Fragile X Syndrome: Parent and Caregiver Perspectives
- Clinical Trials Mini Survey — Survey: Research Experiences in Fragile X Families
- Voice of the People: Treatment Priorities — Voice of People with Fragile X Syndrome and Their Families: Reports from a Survey on Treatment Priorities
ANXIETY SURVEY
Observable Symptoms of Anxiety in Individuals with Fragile X Syndrome: Parent and Caregiver Perspectives
Dates Collected: August–November 2019
Respondents: 518
Ages: 3–70 (mean age 36.5)
*Preliminary numbers.
Sample Data:
FXS: I have Fragile X syndrome
Caretaker: I am a family member or caretaker of someone with FXS
Professional: I am a professional who works with a person with FXS
CLINICAL TRIALS MINI SURVEY
Survey: Research Experiences in Fragile X Families
Dates Collected: September 28–November 2, 2020
Respondents: 225
*Preliminary numbers.
Sample Data:
VOICE OF THE PEOPLE: TREATMENT PRIORITIES
Voice of People with Fragile X Syndrome and Their Families: Reports from a Survey on Treatment Priorities
Dates Collected: October 2017–January 2018
Respondents: 468
*Preliminary numbers.
Sample Data:
FXS: I have Fragile X syndrome
Caretaker: I am a family member or caretaker of someone with FXS
Professional: I am a professional who works with a person with FXS
*Full Question: What is the age of the person with FXS with whom you have the connection? Or your age if you have FXS.
2. NFXF Data Use Policy
Applicants will need to review the information in our Data Use Policy. This document is also available in the application. If approved, you’ll be asked to also sign this document and return it to us.
3. Data Access Application
4. NFXF Data Request Committee Review
The NFXF Data Request Committee will review the Data Access Application for its methodological soundness, anticipated contribution to the community, and lack of overlap with other completed, ongoing, or previously approved planned analyses using the same data. All requests will be reviewed within 4 weeks of submission.
5. Dataset Delivered
If approved, a raw dataset and accompanying documentation will be provided securely to the principal investigator. All data access, use, and storage information is provided in our Data Use Policy document.
About the NFXF Data Repository
The goal of the NFXF Data Repository™ is to advance scientific understanding and treatment research for Fragile X by leveraging previously collected data for new proof-of-concept drug treatments, outcome measure development, and other meaningful scientific work.
Patient data can live on beyond initial clinical trials, creating opportunities for patient samples and clinical data to aid in multiple future treatment discovery programs. Researchers can feel confident knowing that the NFXF Data Repository has quality data from individuals with Fragile X syndrome, and the Fragile X community will know and trust that their data is safely housed within the NFXF.
Through the NFXF Data Repository, the paradigm shifts from the current model — where clinical trial data stays locked up with the sponsor, with no future use for the data — to a new NFXF model — where data is set free to become part of a treatment development cycle.
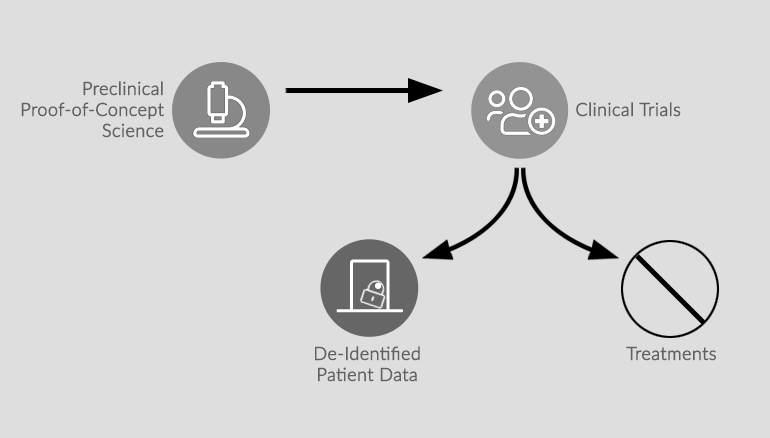
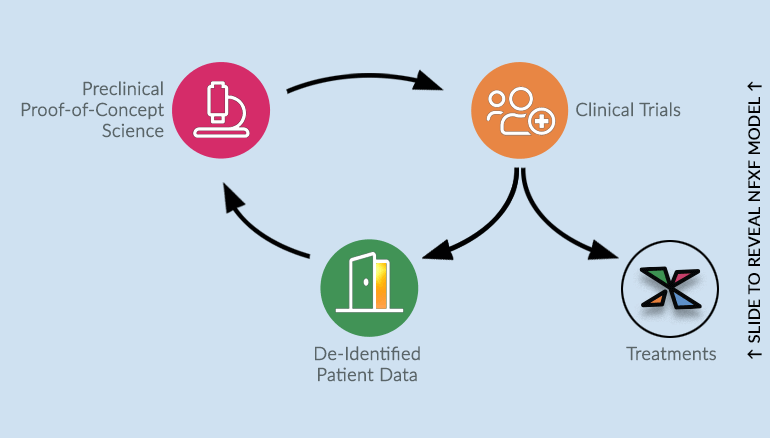
Supporting All Stakeholders
FACT: Multiple clinical trials for treatments specific to Fragile X syndrome have failed, leaving the community searching for treatments to improve quality of life in FXS.
FACT: Clinical trials take an average of 11 years and $2.6 billion to bring a drug to market.
We want to leverage participants’ contributions in clinical trials and make the de-identified data widely available to the Fragile X research community to expedite treatment development. This is part of the NFXF’s effort to support all stakeholders — Fragile X professionals, families, and researchers — to be engaged in the process to promote better outcomes for families living with Fragile X.
Our Role as Steward
There is a growing trend of patient advocacy organizations becoming more actively involved in data stewardship. Given our status as the leading patient advocacy organization for Fragile X, and our experience with collecting data in partnership with the FXCRC clinics and the CDC for the FORWARD registry and database, the NFXF is the best equipped to centralize all de-identified clinical trial data for future research. The NFXF can provide unbiased access to all reputable projects wanting to utilize these data, while at the same time ensuring that all data remains de-identified for maximal patient safety and confidentiality.
NFXF is the best equipped to centralize all de-identified clinical trial data for future research.
Our efforts to advance treatment research ensures that valuable clinical and biological data from clinical trials can live on beyond the initial trials, furthering the investment of time and resources for families and researchers, and ultimately supporting new treatment research.
A Readily Available, Inclusive Repository
The NFXF Data Repository™ is an inclusive repository of all valuable data, including NFXF survey data and NFXF Biobank data. The Data Repository allows research data to be readily available to the broader community for future research.
- Leveraging patient data from previous trials will enable new trialists to complete their proof-of-concept work without having to recruit participants and collect samples.
- Leveraging survey data from prior NFXF work enables development of better outcome measures and serves as a jumping off point for collaborative conversations to better research in Fragile X.
This is important because we know that there are many important things that lead to advancing treatment research. Having data living on past its initial collection is best for families and cost effective for researchers. Both clinical and biological data are valuable, and the Data Repository captures the importance of both. We have multiple sources contributing data, including our NFXF Biobank pilot project, the STX209 Reconsent project, and data rollover from future trials. The paired clinical and biological de-identified data in the Data Repository will allow researchers to jumpstart projects in meaningful ways, including treatment development.
Biomarkers are a big piece of the researcher puzzle, and so are better outcome measures specific to Fragile X and better representation in research so that treatments are effective for any individual living with Fragile X. The NFXF Data Repository allows us to look at research from these angles, doing parallel work in impactful areas. Including qualitative data in the NFXF Data Repository helps us examine additional angles.
The Data Repository includes qualitative survey data, like the Voice of the Person and Anxiety Survey (available next year) data, that can contribute to additional meaningful work to advance treatment research.
We know we need better outcome measures in FX. NFXF plans to start this collaborative conversation with industry and academic researchers by utilizing the survey data in the NFXF Data Repository. Understanding how self-advocates, parents, and caregivers perceive and rate the symptoms of Fragile X is important in developing a measurement that can accurately capture baseline symptomology and change.
why we do what we do

Tino

Jace

Cole and his grandma
Jody and Dayna

Liam and Olivia
Questions?
If you have questions about anything research-related, we’d love to hear from you! You can reach out to Hilary Rosselot directly, or submit your question or comment through our contact form below.

Hilary Rosselot, Director of Research Facilitation
hilary@fragilex.org | (202) 747-6208
Last Updated: 5/9/2023

