Introduction
Families, and in some cases professionals, are often not clear about the relationship between Fragile X syndrome (FXS) and autism spectrum disorder (ASD). It is not uncommon for a child to initially be diagnosed with FXS and later to receive an additional diagnosis of ASD or vice versa.
This document attempts to bring clarity to how the ASD diagnosis and FXS can overlap, and where they do not. Understanding this distinction can be particularly helpful for genetic counseling and when deciding upon the most appropriate medical, therapeutic, educational, and behavioral interventions that will increase the potential for both short-term and long-term improvements. At the end of this document, readers will find general treatment recommendations based on the current expert consensus and evidence-based publications in the field of ASD in FXS.
What is Autism Spectrum Disorder?
ASD is a neurodevelopmental disorder characterized by social communication challenges and the presence of restricted and repetitive behaviors. ASD symptoms usually appear in early childhood and change over time and with development.
The diagnosis of ASD is based on observations and assessments of behavior by an experienced clinician, where clinical judgment is informed by the use of standardized diagnostic instruments and where there is a focus on core social communication and interaction and restricted and repetitive behavior symptoms.[38, 49] The gold standard for clinical diagnosis of ASD in FXS involves the use of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5; American Psychiatric Association, 2013) or the International Classification of Diseases, 11th Edition (ICD-11).
In earlier DSM-based guidelines (DSM-IV), ASD was represented by three different diagnoses: autistic disorder; Asperger syndrome; and pervasive developmental disorder, not otherwise specified (PDD-NOS). These three diagnoses were combined into the one diagnosis of ASD under the DSM-5. The term, “spectrum,” in ASD means that people can be affected in different ways, and symptoms can range from having mild to severe impacts on a person’s functioning.
The symptoms of ASD do not result solely from limited communication skills or intellectual disability (ID), though both may be present. Instead, ASD represents a difficulty in using communication and intellectual abilities for social interaction. ASD involves a number of behaviors, as described in the bullet points below.
Social communication and social interaction symptoms in ASD include challenges with:
- Social-emotional reciprocity — having a conversation, sharing interests and emotions
- Nonverbal communication used for social interaction — differences in use of eye contact, facial expressions, or gestures
- Developing and understanding social relationships — friendships, understanding rules for social behavior
Restricted, repetitive behaviors include:
- Stereotyped or repetitive motor movements — hand flapping, use of objects (e.g., spinning or lining up toys), and speech (using repetitive or unusual words or phrases)
- Insistence on sameness or ritualized patterns of behavior — extreme distress at small changes, difficulties with transitions, needing to follow the same schedule or sequence of completing activities
- Unusual or overly intense interests — interest in an unusual object or topic, intense interest in a narrow area
- Hyper- or hypo-reactivity to sensory input — negative reactions to certain textures or sounds; excessive smelling, peering at, or touching of objects
When diagnosing ASD, the clinician should state whether it is associated with a known medical, genetic, or environmental factor. They should also specify whether ASD is with or without accompanying ID and with or without accompanying language delays.
What is Known About the Cause of ASD?
ASD is a complex condition with a common set of behavioral symptoms, but with varied, yet not well understood, underlying risk factors and biological mechanisms. Although people with ASD share core features of social interaction challenges and restricted, repetitive behaviors, these symptoms can vary widely in form and severity. Further complicating the picture, many individuals with ASD have co-occurring conditions, such as language disorders, ID, or mental health diagnoses (e.g., ADHD, anxiety). Among the causes and risk factors noted by both the U.S. Centers for Disease Control (CDC) and the National Institutes of Health (NIH):
- Research has identified around 100 high-confidence risk genes, but many are yet to be discovered.[18, 41, 83].
- Children who have a sibling with ASD are at a higher risk of also having ASD.[66]
- Individuals with certain genetic or chromosomal conditions, such as FXS, can have a greater chance of having ASD.
It is expected that as genetic testing becomes more sensitive, the percentage of individuals with an identified genetic cause of ASD will increase further.[18] However, at this time, there is no medical test, such as a blood test or brain scan that can diagnose ASD.
What is Known About the Relationship Between FXS and ASD?
FXS is the most common single gene disorder associated with ASD, accounting for about 1-6% of all cases of ASD.[60, 69] FXS is caused by an expansion of a CGG repeat sequence to >200 repeats (full mutation) in the promoter region of the FMR1 gene, which results in a loss of its encoded protein, the Fragile X Messenger Ribonucleoprotein (FMRP). [26]
FXS is diagnosed by a DNA blood test, unlike ASD, which is a purely behaviorally defined diagnosis. The behavioral characteristics of FXS, although quite variable, can include many features of ASD, such as difficulties with social interaction and communication (e.g., poor eye contact, problems with peer relationships, social withdrawal), repetitive movements (e.g., hand flapping), need for sameness, and self-injurious behavior (e.g., hand biting).[10, 25] Thus, careful discernment and a clinical evaluation are needed to understand whether ASD is present in an individual with FXS.
Many individuals diagnosed with FXS meet criteria for a diagnosis of ASD, with the estimated prevalence of ASD in FXS being approximately 50% for males and 20% for females. [44]
However, recent research indicates that, while a majority of males and females with FXS have restricted and repetitive behaviors common to ASD, far fewer meet criteria for the social communication and interaction problems associated with ASD. Receiving a diagnosis of FXS and ASD can be appropriate, but the relationship between FXS and ASD and the characteristics of ASD seen in FXS are still an evolving science and the diagnosis of ASD in FXS continues to be studied by clinicians and researchers working with people with FXS.
Many individuals diagnosed with FXS meet criteria for a diagnosis of ASD, with the estimated prevalence of ASD in FXS being approximately 50% for males and 20% for females.
Why Are Autistic Features Found in FXS?
Our current knowledge about ASD indicates that it is a developmental brain disorder, beginning shortly after birth or even earlier. Its most characteristic feature is the presence of abnormal patterns of neural “wiring” or connectivity. Because multiple genetic and environmental factors have been linked to ASD, there are probably multiple ways in which neural connectivity and other processes can be disrupted, leading to ASD [19,50].
There is evidence, based on studies of genes and proteins in neural cells, that FMRP plays a role in the processes that lead to ASD. FMRP, which is absent or deficient in FXS, normally turns off protein synthesis (the process of proteins being made) at the level of connections between neurons (dendrites). When certain receptors (such as mGluR, dopamine D2, and muscarinic cholinergic) are activated, this releases the FMRP brake and allows proteins to be made. In this way, FMRP regulates the levels of many proteins important at brain connections. Also, many proteins that have similar functions to FMRP, and also interact with FMRP, have been found to be associated with ASD.[14, 15] In addition, many of the proteins regulated by FMRP have been found to be associated with ASD.[3, 41] Thus, deficits in FMRP seem to be linked to abnormal wiring and related brain abnormalities that lead to behavioral symptoms of ASD.
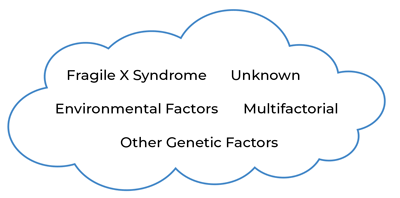
It may be helpful to think of ASD as a “cloud” (see Figure 1), where multiple diverse genetic and other causes lead to atypical brain wiring that results in a final common diagnostic category. Some causes may stem from a single genetic change, some causes may require a number of factors to interact together (i.e., multifactorial), and some causes may be unknown. Although the causes can vary, the end result is the set of behaviors known as ASD.
Individuals with FXS who meet criteria for the current definition of ASD represent an area in the ASD “cloud” where an individual may meet for ASD criteria due to reduced social interactions with others, reduced interest in social interaction, and increased repetitive behaviors in addition to the common characteristics associated with FXS, such as social anxiety, intellectual disability, and sensory hyperreactivity. [27] However, not all individuals with FXS meet criteria for the diagnosis of ASD. FXS is not a kind of ASD; rather, it is a genetic disorder with its own set of symptoms, many of which overlap with behavioral symptoms seen in ASD. When enough overlapping symptoms are present and severe enough to impact functioning, the person with FXS could also meet criteria for a diagnosis of ASD. Some individuals with FXS may have enough ASD features to be near the ASD cloud, but not at the threshold needed for an ASD diagnosis, and those with FXS who are in the ASD cloud likely have genetic factors besides FMRP deficiency that contribute to the development of ASD.
FXS is not a kind of ASD; rather, it is a genetic disorder with its own set of symptoms, many of which overlap with behavioral symptoms seen in ASD.
Intellectual disability is an important variable in considering whether ASD is present within FXS. ID, a feature of FXS in most males, is common in ASD but is not necessary for a diagnosis of ASD, nor for FXS with ASD. However, severe ID is often difficult to differentiate from ASD, as it, too, includes challenges in social development and communication, and repetitive and self-stimulatory behaviors are often present.
The DSM-5 requires consideration of overall intellectual level in determining an ASD diagnosis. If a child’s social communication delays are in line with their overall developmental level, then a diagnosis of ASD is not warranted. In other words, social challenges must exceed those expected based on a person’s overall developmental level.
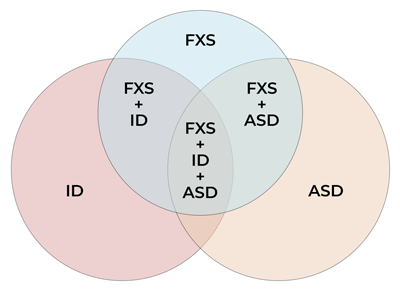
Figure 2 is a Venn diagram used to illustrate the relationship between ASD and FXS but it is not designed to be proportional.
Similarities and Differences
Neurological and behavioral characteristics common to both FXS and ASD include:
- Social interaction problems, including being able to accurately read and respond to social situations
- Communication-language delays
- in FXS involving a wide range of language and speech aspects
- in ASD mainly involving non-verbal communication and language pragmatics (use of language in social situations)
- Poor eye contact (though this often improves in FXS as the person gets to the know the other person)
- Repetitive use of objects and repetitive movements
- Intellectual disability in most males and some females with FXS and a high proportion of those with ASD
- Unusual reactions to sensory input, including hyper-sensitivity (e.g., tactile defensiveness) and hypo-sensitivity (e.g., high tolerance for pain, seeking sensory input such as visual fascination with moving objects or lights), the latter more common in ASD
- An area of strength in visual memory
- Seizures in a higher proportion than the general population
- Motor coordination difficulties (e.g., challenges with handwriting, atypical walking patterns)
- Difficulties with attention, activity level, emotional-behavioral regulation and mood, often leading to additional diagnoses
- Other behavioral issues (e.g., aggression, noncompliance, self-injury)
- Sleep problems
While both ASD and FXS (without ASD) involve social challenges, clinicians working with individuals with FXS observe distinct differences in that lack of social initiations alone do not necessarily imply the absence of social awareness or social interest in individuals with FXS.
For most individuals with FXS, lack of social initiation often reflects language delays, intellectual disabilities, and general or social anxiety.
In contrast, lack of social initiation in ASD, including individuals with FXS who meet criteria for ASD, are defined based on reduced interest in social interaction or a failure to attend to social information that might promote social behavior.
A similar trend is found for eye contact. Although poor eye contact is characteristic of both FXS and ASD, the nature of eye contact may be substantially different.
Individuals with FXS typically avoid eye contact directly , and looking away from people may help in coping with emotional discomfort that is driven by underlying social anxiety. [10, 79, 28]
While social anxiety might also play a role in some individuals with ASD, including those with FXS who meet criteria for ASD, other individuals with ASD make little eye contact for other reasons, such as a lack of recognition that eye gaze is a source of social information or interaction. [32, 35]
Characteristics that have been found to differ between FXS and ASD include:
- Individuals with ASD typically have higher levels of motor coordination.
- Individuals with ASD are more likely to show higher expressive language skills relative to receptive language, while individuals with FXS tend to show higher receptive language skills relative to expressive [1]
- In general, interest in socializing is higher in FXS than ASD, though anxiety and language delays can be limiting factors [17, 62]
- Social awareness is typically better in FXS than ASD
- Motor imitation skills are typically better in FXS than ASD [51, 53]
- Sequential processing is typically harder for individuals with FXS than ASD [33]
Characteristics of Individuals with FXS with ASD
Outlined above were descriptions of behaviors that are common and different between FXS and ASD. Compared to individuals with FXS who do not meet criteria for ASD, those who do meet ASD criteria present with more severe cognitive and behavioral problems that include:
- Less developed language skills, particularly receptive skills [1, 57]
- Often non-verbal or minimally verbal [57]
- Lower IQ scores [9, 11, 42, 62]
- Lower adaptive skills [9, 42]
- More severe overall behavioral problems, including attention problems, hyperactivity and impulsivity, anxiety (particularly after childhood), irritability, aggression, agitation, self-injury, hypersensitivity to stimuli, and obsessive-compulsive and perseverative behavior [9, 16, 44]
- Sleep problems, particularly after childhood [44]
- Seizures [44]
It is interesting to note that the above list contains characteristics that are not core features of ASD. Rather, they reflect common comorbidities and greater developmental impairment. It is important to note that many of these characteristics are seen in individuals with FXS with significant ID, who do not have ASD. Therefore, the diagnosis of ASD can be difficult to make in these individuals with FXS and significant ID. So, when making the ASD diagnosis in FXS, consideration must be given to the cognitive level of the individual to ensure that ASD symptoms (social communication challenges and repetitive behaviors) are more impaired than expected based on overall developmental level.
Future studies are needed to improve methods for diagnosis of ASD in those with ID. Using data from the FORWARD project (A CDC-funded, multi-year, natural history study involving multiple Fragile X clinics), revised questionnaires for the diagnosis of ASD that are more appropriate for FXS have been developed, and further work is needed to refine ASD diagnostic measures for use in individuals with FXS.[45]
From the educational, vocational, and adaptive functioning viewpoints, individuals with FXS with ASD face similar but more severe challenges than those with FXS without ASD. Individuals with FXS with ASD have a higher frequency of use of medications for behavioral management, as well as higher likelihood of having very delayed toilet training, seizures, and sleep problems [7, 16, 44].
Failure to accurately identify ASD in FXS has multiple implications including that developmental and neurobehavioral comorbidities associated with ASD in FXS may not be recognized, leading to failure to enact preventive strategies. An inadequate diagnosis may also prevent the implementation of interventions and educational strategies shown to have benefit in ASD, such as applied behavior analysis (ABA). More precise delineation of ASD behaviors exhibited by an individual with FXS could also assist in developing targeted interventions.[44] Therefore, accurate and early diagnosis and prompt referral of children with FXS and ASD to receive appropriate therapies and supports is essential to make an early and positive impact on outcomes.
Closing
Up to half of the people with FXS will also have a behavioral diagnosis of ASD, and it can be useful to get the dual diagnosis early as it may impact treatment and intervention. It is important to note that, while many people with FXS with ASD will have increased challenges, they may also have the positive attributes of those with FXS without ASD including having a good sense of humor, a desire to engage with others, and to be helpful. It is important to use those strengths to further their academic and life skills. Regardless of ASD status, evaluations and treatment plans work best when they are individualized to an individual’s specific needs and profile of strengths and weaknesses.


